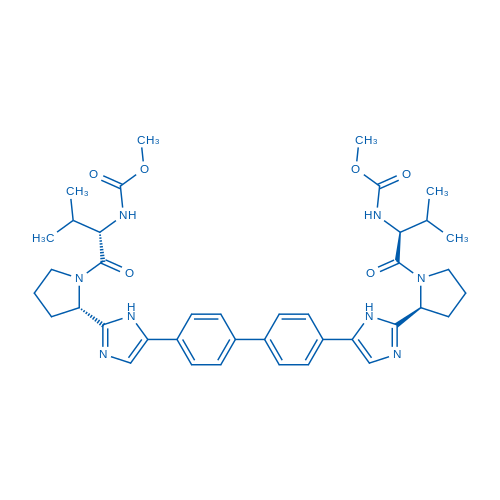
CAS No.: 1009119-64-5
达拉他韦 Catalog No. CSN12990
Synonyms: BMS-790052;EBP 883;Daklinza
Daclatasvir is a first-in-class, highly-selective oral HCV NS5A inhibitor.
纯度 & 质量文件
批次:
靶点选择性
生物活性
- 描述
- 细胞研究
- Cell Data
- 更多
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT02095860 Hepatitis C Phase 1 Completed - United States, Texas ... more >> Healthcare Discoveries, Llc D/B/A Icon Development Solutions San Antonio, Texas, United States, 78209 Collapse << NCT02531269 - - Completed - Switzerland ... more >> Local Institution Basel, Switzerland Collapse << NCT03186313 Hepatitis C Phase 3 Completed - Egypt ... more >> Egyptian Liver Hospital Shirbīn, Dakahlia, Egypt, 35681 Collapse << - 更多
- 参考文献
- [1] Lee C, Ma H, et al. The hepatitis C virus NS5A inhibitor (BMS-790052) alters the subcellular localization of the NS5A non-structural viral protein. Virology. 2011 May 25;414(1):10-8.
- [2] Gao M, Nettles RE, et al. Chemical genetics strategy identifies an HCV NS5A inhibitor with a potent clinical effect. Nature. 2010 May 6;465(7294):96-100.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1009119-64-5 | 储存条件 |
|
|||||
| 分子式 | C40H50N8O6 | 运输 | 蓝冰 | |||||
| 分子量 | 738.88 | 别名 | BMS-790052;EBP 883;Daklinza;UNII-LI2427F9CI;达拉他韦 | |||||
| 溶解度 |
|
动物实验配方 |
| Cell Lines | Concentration | Assay Type | Time | Activity Description | Data Sources |
|---|
| african green monkey Vero cells | - | Cytotoxicity assay | - | Cytotoxicity against african green monkey Vero cells, CC50=9.6 μM | 23466233 |
| human CEM cells | - | Cytotoxicity assay | 3 days | Cytotoxicity against human CEM cells after 3 days, CC50=9.6 μM | 25154714 |
| human CEM cells | - | Cytotoxicity assay | - | Cytotoxicity against human CEM cells, CC50=9.6 μM | 22704887 |
| human Huh7 cells | - | Function assay | 3 days | Antiviral activity against HCV genotype 1b infected in human Huh7 cells after 3 days by cell-based replicon assay, EC50=3e-06 μM | 25148100 |
| human PBMC cells | - | Cytotoxicity assay | - | Cytotoxicity against human PBMC cells, CC50=19 μM | 22704887 |
| NCT号 | 适应症或疾病 | 临床期 | 招募状态 | 预计完成时间 | 地点 |
|---|
| NCT02095860 | Hepatitis C | Phase 1 | Completed | - | United States, Texas ... more >> Healthcare Discoveries, Llc D/B/A Icon Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT02531269 | - | - | Completed | - | Switzerland ... more >> Local Institution Basel, Switzerland Collapse << |
| NCT03186313 | Hepatitis C | Phase 3 | Completed | - | Egypt ... more >> Egyptian Liver Hospital Shirbīn, Dakahlia, Egypt, 35681 Collapse << |
| NCT02170727 | Hepatitis C Virus | Phase 3 | Completed | - | Korea, Republic of ... more >> Local Institution Busan, Korea, Republic of, 602-739 Local Institution Busan, Korea, Republic of, 614-735 Local Institution Gyeonggi-do, Korea, Republic of, 463-707 Local Institution Gyeonggi-Do, Korea, Republic of, 480-717 Local Institution Gyeongsangnam-do, Korea, Republic of, 626-770 Local Institution Inchoen, Korea, Republic of, 405-760 Local Institution Seoul, Korea, Republic of, 120-752 Local Institution Seoul, Korea, Republic of, 135-710 Local Institution Seoul, Korea, Republic of, 138-736 Local Institution Seoul, Korea, Republic of, 156-755 Russian Federation Local Institution Kazan, Russian Federation, 420140 Local Institution Moscow, Russian Federation, 109240 Taiwan Local Institution Kaohsiung, Taiwan, 807 Local Institution Kaohsiung, Taiwan, 833 Local Institution Taichung, Taiwan, 40447 Local Institution Taichung, Taiwan, 40705 Local Institution Tainan, Taiwan, 704 Local Institution Taipei, Taiwan, 100 Local Institution Taipei, Taiwan, 112 Local Institution Taoyuan, Taiwan, 333 Collapse << |
| NCT02045966 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT01016912 | Hepatitis C Infection | Phase 2 | Completed | - | Japan ... more >> Local Institution Hiroshima City, Hiroshima, Japan, 734-0037 Local Institution Sapporo-Shi, Hokkaido, Japan, 060-0033 Local Institution Kawasaki-Shi, Kanagawa, Japan, 2138587 Local Institution Suita-Shi, Osaka, Japan, 5650871 Local Institution Iruma-Gun, Saitama, Japan, 3500495 Local Institution Minato-Ku, Tokyo, Japan, 105-0001 Collapse << |
| NCT01573351 | Hepatitis C Virus | Phase 3 | Completed | - | - |
| NCT02727933 | - | - | Recruiting | September 30, 2020 | Korea, Republic of ... more >> Local Institution Recruiting Seoul, Korea, Republic of Contact: Site 0001 Collapse << |
| NCT02250001 | - | - | Completed | - | Japan ... more >> Local Institution Chuo-ku, Tokyo, Japan, 104-0033 Collapse << |
| NCT01016912 | - | - | Completed | - | - |
| NCT02292966 | Hepatitis C, Chronic | Phase 4 | Withdrawn | June 2016 | Australia, New South Wales ... more >> St Vincent's Hospital Sydney, New South Wales, Australia, 2010 Westmead Hospital Westmead, New South Wales, Australia, 2145 Collapse << |
| NCT03500562 | - | - | Recruiting | December 31, 2021 | China, Beijing ... more >> Local Institution Recruiting Beijing, Beijing, China, 100054 Contact: Site 0002 Collapse << |
| NCT01741545 | Hepatitis C Virus | Phase 3 | Completed | - | - |
| NCT00983957 | - | - | Completed | - | - |
| NCT02161939 | - | - | - | - | - |
| NCT00983957 | Chronic Hepatitis C | Phase 1 | Completed | - | United States, Arizona ... more >> MDS Pharma Services (US), Inc Tempe, Arizona, United States, 85283 United States, Texas Covance Clinical Research Unit, Inc. Austin, Texas, United States, 78752 Canada, Quebec Local Institution St. Laurent, Quebec, Canada, H4R2N6 Collapse << |
| NCT02175966 | Hepatitis C | Phase 2 | Completed | - | United States, California ... more >> Inland Empire Liver Foundation Rialto, California, United States, 92377 United States, Illinois Northwestern University Feinberg School Of Medicine Chicago, Illinois, United States, 60611 United States, Indiana Indiana University Health - University Hospital Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins University Lutherville, Maryland, United States, 21093 United States, Texas Texas Liver Institute San Antonio, Texas, United States, 78215 Collapse << |
| NCT00874770 | Hepatitis C Infection | Phase 2 | Completed | - | United States, Alabama ... more >> Alabama Liver & Digestive Specialists (Alds) Montgomery, Alabama, United States, 36116 United States, Colorado University Of Colorado Denver & Hospital Aurora, Colorado, United States, 80045 United States, Connecticut Yale University School Of Medicine New Haven, Connecticut, United States, 06520 United States, Maryland Mercy Medical Center Baltimore, Maryland, United States, 21202 United States, Massachusetts Llc Dba The Research Institute Springfield, Massachusetts, United States, 01107 United States, New York Veterans Affairs Medical Center Bronx, New York, United States, 10468 United States, North Carolina Carolinas Center For Liver Disease Statesville, North Carolina, United States, 28677 United States, Oklahoma Options Health Research, Llc Tulsa, Oklahoma, United States, 74104 Healthcare Research Consultants Tulsa, Oklahoma, United States, 74135 United States, Texas North Texas Research Institute Arlington, Texas, United States, 76012 United States, Virginia Metropolitan Research Fairfax, Virginia, United States, 22031 France Local Institution Creteil, France, 94010 Local Institution Paris Cedex 14, France, 75679 Local Institution Vandoeuvre Les Nancy, France, 54511 Collapse << |
| NCT00874770 | - | - | Completed | - | - |
| NCT01359644 | Chronic Hepatitis C | Phase 2 | Completed | - | United States, California ... more >> Southern California Liver Centers Coronado, California, United States, 92118 Research And Education, Inc. San Diego, California, United States, 92105 United States, Colorado University Of Colorado Denver & Hospital Aurora, Colorado, United States, 80045 United States, Florida University Of Florida Hepatology Gainesville, Florida, United States, 32610 Orlando Immunology Center Orlando, Florida, United States, 32803 Miami Research Associates South Miami, Florida, United States, 33143 United States, Maryland Mercy Medical Center Baltimore, Maryland, United States, 21202 Johns Hopkins University Lutherville, Maryland, United States, 21093 United States, Michigan University Of Michigan Health System Ann Arbor, Michigan, United States, 48109 United States, New York Bronx Va Medical Center 3c Sub-J Bronx, New York, United States, 10468 Weill Cornell Medical College New York, New York, United States, 10021 United States, Oklahoma Options Health Research, Llc Tulsa, Oklahoma, United States, 74104 Healthcare Research Consultants Tulsa, Oklahoma, United States, 74135 United States, Pennsylvania University Of Pennsylvania Philadelphia, Pennsylvania, United States, 19104 United States, Texas Alamo Medical Research San Antonio, Texas, United States, 78215 United States, Virginia Metropolitan Research Annandale, Virginia, United States, 22003 United States, Wisconsin Dean Clinic Madison, Wisconsin, United States, 53715 Puerto Rico Local Institution San Juan, Puerto Rico, 00927 Collapse << |
| NCT00859053 | Hepatic Insufficiency | Phase 1 | Completed | - | United States, California ... more >> Advanced Clinical Research Institute Anaheim, California, United States, 92801 United States, Florida Orlando Clinical Research Center Orlando, Florida, United States, 32809 Collapse << |
| NCT01448044 | Hepatitis C | Phase 3 | Completed | - | United States, California ... more >> Scti Research Foundation San Clemente, California, United States, 92673 United States, Massachusetts Umass Memorial Medical Center Worcester, Massachusetts, United States, 01655 United States, Rhode Island University Gastroenterology Providence, Rhode Island, United States, 02905 United States, Virginia Metropolitan Research Annandale, Virginia, United States, 22003 France Local Institution Bondy Cedex, France, 93143 Local Institution Creteil, France, 94000 Local Institution La Roche-Sur-Yon Cedex 9, France, 85925 Local Institution Marseille Cedex 08, France, 13285 Local Institution Nice Cedex 03, France, 06202 Local Institution Orleans Cedex 2, France, 45067 Local Institution Paris, France, 75013 Local Institution Paris, France, 75475 Local Institution Strasbourg Cedex, France, 67091 Local Institution Toulouse Cedex 09, France, 31059 Local Institution Villejuif, France, 94804 Greece Local Institution Thesaloniki, Greece, 54639 Italy Local Institution Roma, Italy, 00149 Local Institution Torino, Italy, 10126 Puerto Rico Local Institution San Juan, Puerto Rico, 00927 Spain Local Institution A Coruna, Spain, 15706 Local Institution Barcelona, Spain, 08003 Local Institution Barcelona, Spain, 08035 Local Institution Madrid, Spain, 28046 United Kingdom Local Institution London, Greater London, United Kingdom, SE5 9RS Local Institution London, Greater London, United Kingdom, SW17 0QT Local Institution London, Greater London, United Kingdom, W2 1NY Collapse << |
| NCT01359644 | - | - | Completed | - | - |
| NCT02103569 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT00859053 | - | - | Completed | - | - |
| NCT01795911 | Hepatitis C | Phase 2 | Completed | - | United States, Texas ... more >> Alamo Medical Research San Antonio, Texas, United States, 78215 Collapse << |
| NCT01497834 | Hepatitis C | Phase 3 | Completed | - | Japan ... more >> Local Institution Nagoya-shi, Aichi, Japan, 4668560 Local Institution Chiba-shi, Chiba, Japan, 2608677 Local Institution Fukuoka-shi, Fukuoka, Japan, 8108563 Local Institution Kurume, Fukuoka, Japan, 8300011 Local Institution Ogaki-shi, Gifu, Japan, 5038502 Local Institution Hiroshima-shi, Hiroshima, Japan, 7340037 Local Institution Sapporo-Shi, Hokkaido, Japan, 0600033 Local Institution Amagasaki-shi, Hyogo, Japan, 6608511 Local Institution Kanazawa-shi, Ishikawa, Japan, 9208641 Local Institution Takamatsu-shi, Kagawa, Japan, 7608557 Local Institution Kagoshima-shi, Kagoshima, Japan, 8908520 Local Institution Kawasaki-Shi, Kanagawa, Japan, 2138587 Local Institution Sendai-Shi, Miyagi, Japan, 9808574 Local Institution Okayama-shi, Okayama, Japan, 7008558 Local Institution Osaka-sayama-shi, Osaka, Japan, 5898511 Local Institution Osaka-shi, Osaka, Japan, 5458586 Local Institution Suita-shi, Osaka, Japan, 5650871 Local Institution Suita, Osaka, Japan, 5640013 Local Institution Iruma-Gun, Saitama, Japan, 350-0495 Local Institution Bunkyo-Ku, Tokyo, Japan, 1138655 Local Institution Minato-ku, Tokyo, Japan, 1058470 Local Institution Musashino-shi, Tokyo, Japan, 1808610 Local Institution Shinagawa-ku, Tokyo, Japan, 1428666 Local Institution Chuo-shi, Yamanashi, Japan, 4093898 Collapse << |
| NCT01428063 | Hepatitis C Virus Infection | Phase 2 | Completed | - | - |
| NCT01448044 | - | - | Completed | - | - |
| NCT01017575 | - | - | Completed | - | - |
| NCT01471574 | Hepatitis C, Genotype 1 | Phase 3 | Completed | - | - |
| NCT01428063 | - | - | Completed | - | - |
| NCT03181074 | - | - | Recruiting | April 11, 2019 | Mexico ... more >> Local Institution Not yet recruiting Benito Juarez, Distrito Federal, Mexico, 03900 Contact: Site 0002 Local Institution Recruiting Mexico, D.f., Distrito Federal, Mexico, 14050 Contact: Site 0001 Collapse << |
| NCT01017575 | Hepatitis C Infection | Phase 2 | Completed | - | Japan ... more >> Local Institution Chiba-Shi, Chiba, Japan Local Institution Kurume-Shi, Fukuoka, Japan, 8300011 Local Institution Okayama-Shi, Okayama, Japan, 7008558 Local Institution Osaka-Shi, Osaka, Japan, 5438555 Local Institution Osaka-Shi, Osaka, Japan, 545-8586 Local Institution Musashino-Shi, Tokyo, Japan, 180-0023 Collapse << |
| NCT02673489 | Hepatitis C | Phase 3 | Completed | - | United States, California ... more >> Keck Medical Center Of USC Los Angeles, California, United States, 90033 University Of California, San Francisco San Francisco, California, United States, 94143 United States, Georgia Gastrointestinal Specialists of Georgia, PC Marietta, Georgia, United States, 30060 United States, Illinois Ruth Rothstein Core Center Chicago, Illinois, United States, 60612 United States, Maryland Digestive Disease Associates, PA Catonsville, Maryland, United States, 21228 United States, Pennsylvania Northeast Clinical Research Center Bethlehem, Pennsylvania, United States, 18017 United States, Rhode Island University Gastroenterology Providence, Rhode Island, United States, 02905 United States, Texas Texas Clinical Research Institute Arlington, Texas, United States, 76012 Methodist Transplant Physicians Dallas, Texas, United States, 75203 The Texas Liver Institute San Antonio, Texas, United States, 78215 United States, Virginia Inova Fairfax Hospital Falls Church, Virginia, United States, 22042 Bon Secours St. Mary's Hospital of Richmond, Inc Richmond, Virginia, United States, 23226 Canada, Alberta Local Institution Calgary, Alberta, Canada, T2N 4Z6 Local Institution Edmonton, Alberta, Canada, T6G 2P4 Canada, British Columbia Local Institution Vancouver, British Columbia, Canada, V6Z 2K5 Local Institution Victoria, British Columbia, Canada, V8V 3P9 Canada, Ontario Local Institution Toronto, Ontario, Canada, M5G 2C4 Canada, Quebec Local Institution Montreal, Quebec, Canada, H3T 1E2 Canada, Saskatchewan Local Institution Regina, Saskatchewan, Canada, S4O 0W5 Collapse << |
| NCT02368522 | - | - | Completed | - | Germany ... more >> Local Institution Hamburg, Germany, 20099 Collapse << |
| NCT02673489 | - | - | Completed | - | - |
| NCT01629732 | Hepatitis C Virus | Phase 2 | Withdrawn | June 2014 | United States, Florida ... more >> Local Institution Orlando, Florida, United States, 32804 Collapse << |
| NCT01125189 | Hepatitis C Virus | Phase 2 | Completed | - | - |
| NCT02762448 | - | - | Withdrawn(no patient enrollmen... more >>t) Collapse << | - | - |
| NCT02639585 | Hepatitis C, Chronic | Phase 4 | Unknown | June 2017 | Korea, Republic of ... more >> Pusan National University Hospital Recruiting Busan, Korea, Republic of, 49241 Contact: Won Lim, M.S. +82-51-240-7869 ezmagic@pusan.ac.kr Pusan National University Yangsan Hospital Recruiting Yangsan, Korea, Republic of, 50612 Contact: Young Mi Hong, M.S. 82-55-360-1570 00gurum@hanmail.net Collapse << |
| NCT01471574 | - | - | Completed | - | - |
| NCT01425970 | Hepatitis C | Phase 2 | Terminated(Termination of stud... more >>y was due to safety reasons) Collapse << | - | United States, California ... more >> Acri Phase One Anaheim, California, United States, 92801 Scripps Health Dba Scripps Clinical Research Services La Jolla, California, United States, 92037 Quest Clinical Research San Francisco, California, United States, 94115 United States, Missouri Kansas City Cancer Centers, Llc. Kansas City, Missouri, United States, 64111 United States, Oklahoma Options Health Research, Llc Tulsa, Oklahoma, United States, 74104 United States, Texas Alamo Medical Research San Antonio, Texas, United States, 78215 Collapse << |
| NCT00546715 | - | - | Completed | - | - |
| NCT01389323 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT00546715 | Chronic Hepatitis C | Phase 1 Phase 2 | Completed | - | United States, California ... more >> Advanced Clinical Research Institute Anaheim, California, United States, 92801 United States, Florida Orlando Clinical Research Center Orlando, Florida, United States, 32809 United States, Maryland Parexel International Corporation Baltimore, Maryland, United States, 21225 United States, Minnesota Mayo Clinic Rochester, Minnesota, United States, 55905 United States, New Jersey Bristol-Myers Squibb Clinical Pharmacology Unit Hamilton, New Jersey, United States, 08690 United States, Pennsylvania Thomas Jefferson University Philadelphia, Pennsylvania, United States, 19107 United States, Virginia University Of Virginia Digestive Health Center Of Excellence Charlottesville, Virginia, United States, 22908 Collapse << |
| NCT01125189 | - | - | Completed | - | - |
| NCT01389323 | - | - | Completed | - | - |
| NCT01938625 | Hepatitis C, Chronic | Phase 2 | Completed | - | Germany ... more >> Essen, Germany Hamburg, Germany Poland Warszawa, Poland Spain Barcelona, Spain Madrid, Spain Valencia, Spain Collapse << |
| NCT01938625 | - | - | Completed | - | - |
| NCT03549832 | HCV Coinfection | Not Applicable | Recruiting | January 2019 | Egypt ... more >> Assiut University Hopsital Recruiting Assiut, Egypt, 71515 Contact: Mohamed A Mekky, MD Collapse << |
| NCT01866930 | Chronic Hepatitis C Infection | Phase 3 | Completed | - | - |
| NCT01616524 | Hepatitis C Virus (HCV) | Phase 3 | Completed | - | - |
| NCT01718158 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT02323594 | Hepatitis C Infection | Phase 1 | Completed | - | - |
| NCT01492426 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT00663208 | Chronic Hepatitis C | Phase 2 | Completed | - | United States, California ... more >> Advanced Clinical Res Inst Anaheim, California, United States, 92801 West Coast Clinical Trials, Llc Cypress, California, United States, 90630 United States, Connecticut Yale University School of Medicine New Haven, Connecticut, United States, 06510 United States, Florida Elite Research Institute Miami, Florida, United States, 33169 Orlando Clinical Research Center Orlando, Florida, United States, 32809 United States, Maryland Parexel International Corporation Baltimore, Maryland, United States, 21225 United States, Texas Alamo Medical Research San Antonio, Texas, United States, 78215 Puerto Rico Local Institution Santurce, Puerto Rico, 00909 Collapse << |
| NCT03208322 | - | - | Not yet recruiting | October 31, 2019 | Mexico ... more >> Local Institution Not yet recruiting Df, Distrito Federal, Mexico, 10700 Contact: Site 0002 Local Institution Not yet recruiting Magdalena De Las Salinas, Distrito Federal, Mexico, 07760 Contact: Site 0001 Collapse << |
| NCT01718145 | Hepatitis C Virus Infection | Phase 3 | Completed | - | Japan ... more >> Local Institution Nagoya-shi, Aichi, Japan, 4668560 Local Institution Toyoake Shi, Aichi, Japan, 4701192 Local Institution Chiba-shi, Chiba, Japan, 2608677 Local Institution Fukuoka-shi, Fukuoka, Japan, 8108563 Local Institution Kurume, Fukuoka, Japan, 8300011 Local Institution Ogaki-shi, Gifu, Japan, 5038502 Local Institution Takasaki City, Gunma, Japan, 3700829 Local Institution Hiroshima-shi, Hiroshima, Japan, 7340037 Local Institution Obihiro-shi, Hokkaido, Japan, 080-0016 Local Institution Sapporo-shi, Hokkaido, Japan, 0600033 Local Institution Sapporo-shi, Hokkaido, Japan, 0608648 Local Institution Takamatsu-shi, Kagawa, Japan, 760-8557 Local Institution Kagoshima-shi, Kagoshima, Japan, 8908520 Local Institution Kawasaki-shi, Kanagawa, Japan, 2138587 Local Institution Yokohama-shi, Kanagawa, Japan, 2360004 Local Institution Kumamoto-shi, Kumamoto, Japan, 8608556 Local Institution Kyoto-shi, Kyoto, Japan, 6028566 Local Institution Sendai-shi, Miyagi, Japan, 9808574 Local Institution Matsumoto, Nagano, Japan, 3908621 Local Institution Nagasaki-shi, Nagasaki, Japan, 8528501 Local Institution Omura, Nagasaki, Japan, 8568562 Local Institution Kashihara, Nara, Japan, 6348522 Local Institution Yufu, Oita, Japan, 8795593 Local Institution Okayama-shi, Okayama, Japan, 7008558 Local Institution Osaka-sayama-shi, Osaka, Japan, 5898511 Local Institution Osaka-shi, Osaka, Japan, 5438555 Local Institution Osaka-shi, Osaka, Japan, 5458586 Local Institution Suita-shi, Osaka, Japan, 5650871 Local Institution Suita, Osaka, Japan, 5640013 Local Institution Iruma-gun, Saitama, Japan, 3500495 Local Institution Izunokuni, Shizuoka, Japan, 4102295 Local Institution Shimotsuke-shi, Tochigi, Japan, 3290498 Local Institution Bunkyo-ku, Tokyo, Japan, 1138519 Local Institution Bunkyo-ku, Tokyo, Japan, 1138655 Local Institution Minato-ku, Tokyo, Japan, 1058470 Local Institution Musashino-shi, Tokyo, Japan, 1808610 Local Institution Shinagawa-ku, Tokyo, Japan, 1428666 Local Institution Yamagata-shi, Yamagata, Japan, 9909585 Local Institution Chuo-shi, Yamanashi, Japan, 4093898 Local Institution Fukui, Japan, 9188503 Local Institution Fukuoka, Japan, 8140180 Local Institution Fukuoka, Japan, 8158555 Local Institution Gifu, Japan, 5008513 Local Institution Kumamoto, Japan, 8628655 Local Institution Nagoya-shi, Japan, 4678602 Local Institution Nishinomiya-shi, Japan, 6638501 Local Institution Osaka, Japan, 5400006 Local Institution Saitama, Japan, 3380001 Collapse << |
| NCT02473211 | Chronic Hepatitis C Infection | Phase 2 Phase 3 | Completed | - | China, Beijing ... more >> Liver Fibrosis Diagnosis and Treatment Centre, 302 Hospital Beijing, Beijing, China, 100039 China, Hong Kong Humanity and Health GI and Liver Centre Hong Kong, Hong Kong, China, 00852 Collapse << |
| NCT03748745 | Drug Interactions | Phase 1 | Active, not recruiting | January 19, 2019 | China, Jilin ... more >> Phase I Clinical Trial Unit, The First Hospital of Jilin University Changchun, Jilin, China, 130000 Collapse << |
| NCT03080415 | Hepatitis C Genotype 4 | Phase 3 | Completed | - | Egypt ... more >> Professor Yassin Abdel Ghaffar Charity Center for Liver Disease and Research Nasr City, Cairo, Egypt Collapse << |
| NCT02104843 | Hepatitis C | Phase 1 | Completed | - | - |
| NCT01492426 | - | - | Completed | - | - |
| NCT01725542 | HCV-HIV Co-Infection | Phase 2 | Completed | - | France ... more >> All the Regions of the Country (33 Centers), France Collapse << |
| NCT02349048 | Hepatitis C Virus | Phase 2 | Completed | - | United States, California ... more >> Bakersfield, California, United States United States, Florida Jacksonville, Florida, United States United States, Maryland Lutherville, Maryland, United States United States, North Carolina Winston Salem, North Carolina, United States United States, Tennessee Knoxville, Tennessee, United States United States, Texas Arlington, Texas, United States San Antonio, Texas, United States Canada, Ontario Toronto, Ontario, Canada Collapse << |
| NCT02097966 | - | - | - | - | - |
| NCT02349048 | - | - | Completed | - | - |
| NCT01995266 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT02304159 | Hepatitis C C... more >>irrhosis Collapse << | Phase 4 | Active, not recruiting | December 2017 | United States, California ... more >> Southern California Research Center Coronado, California, United States, 92118 Collapse << |
| NCT03366610 | - | - | Recruiting | April 30, 2024 | China, Beijing ... more >> Local Institution Recruiting Beijing, Beijing, China Contact: Site 0001 Collapse << |
| NCT03205618 | - | - | Completed | - | Saudi Arabia ... more >> Local Institution Doha, Saudi Arabia Local Institution Jeddah, Saudi Arabia, 23325 Local Institution Riyadh-11211, Saudi Arabia, 11211 Collapse << |
| NCT02098616 | Hepatitis C | Not Applicable | Completed | - | United States, California ... more >> VA Long Beach Healthcare System Long Beach, California, United States, 90822 Collapse << |
| NCT03572140 | - | - | Not yet recruiting | August 1, 2020 | - |
| NCT00663208 | - | - | Completed | - | - |
| NCT02565888 | Hepatitis C H... more >>IV Collapse << | Phase 1 | Completed | - | Netherlands ... more >> CRCN, Radboud University Medical Center Nijmegen, Netherlands Collapse << |
| NCT02107365 | Hepatitis C Virus Genotype 4 I... more >>nfection Collapse << | Phase 2 | Completed | - | France ... more >> Hôpital AVICENNE Bobigny, France, 93009 Hôpital Jean Verdier Bondy, France, 93140 Hôpital de Haut Lévêque Bordeaux Pessac, France, 33601 Hôpital Beaujon Clichy, France, 92110 Centre Hospitalier Intercommunal Créteil, France, 94010 Hôpital Henri Mondor Créteil, France, 94010 Hôpital Albert Michallon Grenoble, France, 38043 Hôpital Claude Huriez Lille, France, 59037 Hôpital Dupuytren Limoges, France, 87042 Hôpital de la Croix Rousse Lyon, France, 69317 Fondation Hôpital Saint Joseph Marseille, France, 13285 Hôpital Saint Eloi Montpellier, France, 34295 Hôpital de Brabois Nancy, France, 54511 Hôpital de l'Hôtel Dieu Nantes, France, 44093 Hôpital de l'Archet Nice, France, 06202 Hôpital de La Source Orléans, France Hôpital Saint Antoine Paris, France, 75571 Hôpital Pitié Salpêtrière Paris, France, 75651 Hôpital Cochin Paris, France, 75679 Hôpital Tenon Paris, France, 75970 Hôpital Pontchaillou Rennes, France, 35000 Hôpital Charles Nicolle Rouen, France, 76031 Institut Arnault Tzank Saint Laurent du Var, France, 06721 Hôpital Purpan Toulouse, France, 31059 Hôpital Paul Brousse Villejuif, France, 94804 Collapse << |
| NCT02319031 | Hepatitis C | Phase 3 | Completed | - | Australia, New South Wales ... more >> Local Institution Darlinghurst, New South Wales, Australia, 2010 Australia, Queensland Local Institution Greenslopes, Queensland, Australia, 4120 Australia, South Australia Local Institution Adelaide, South Australia, Australia, 5000 Australia, Victoria Local Institution Clayton, Victoria, Australia, 3168 Local Institution Fitzroy, Victoria, Australia, 3065 Local Institution Heidelberg, Victoria, Australia, 3084 France Local Institution Creteil Cedex, France, 94010 Local Institution Grenoble Cedex 09, France, 38043 Local Institution Paris Cedex 14, France, 75679 Local Institution Vandoeuvre Les Nancy, France, 54511 Collapse << |
| NCT01797848 | Hepatitis C | Phase 3 | Withdrawn | November 2016 | - |
| NCT02319031 | - | - | Completed | - | - |
| NCT01628692 | - | - | Completed | - | - |
| NCT02123654 | Hepatitis C Virus Infection | Phase 3 | Completed | - | Japan ... more >> Local Institution Nagoya-shi, Aichi, Japan, 466-8560 Local Institution Nagoya-shi, Aichi, Japan, 4678602 Local Institution Fukuoka-shi, Fukuoka, Japan, 8108563 Local Institution Kurume-shi, Fukuoka, Japan, 8300011 Local Institution Gifu-shi, Gifu, Japan, 5008513 Local Institution Ogaki-shi, Gifu, Japan, 5038502 Local Institution Takasaki, Gunma, Japan, 3700829 Local Institution Hiroshima-Shi, Hiroshima, Japan, 7348551 Local Institution Sapporo-shi, Hokkaido, Japan, 0600033 Local Institution Sapporo-shi, Hokkaido, Japan, 0608648 Local Institution Kobe-shi, Hyogo, Japan, 6500047 Local Institution Kanazawa-shi, Ishikawa, Japan, 9208641 Local Institution Takamatsu-shi, Kagawa, Japan, 7608557 Local Institution Kagoshima-shi, Kagoshima, Japan, 8908520 Local Institution Kawasaki-shi, Kanagawa, Japan, 2138587 Local Institution Yokohama, Kanagawa, Japan, 2320024 Local Institution Kyoto-shi, Kyoto, Japan, 6028566 Local Institution Kashihara, Nara, Japan, 6348522 Local Institution Okayama-shi, Okayama, Japan, 7008558 Local Institution Osaka-shi, Osaka, Japan, 5438555 Local Institution Osaka-shi, Osaka, Japan, 5458586 Local Institution Suita-shi, Osaka, Japan, 5650871 Local Institution Suita, Osaka, Japan, 5640013 Local Institution Iruma-gun, Saitama, Japan, 3500495 Local Institution Bunkyo-ku, Tokyo, Japan, 1138655 Local Institution Minato-ku, Tokyo, Japan, 1058470 Local Institution Musashino-shi, Tokyo, Japan, 1808610 Local Institution Shinjuku-Ku, Tokyo, Japan, 1608582 Local Institution Yamagata-shi, Yamagata, Japan, 9909585 Local Institution Chuo-shi, Yamanashi, Japan, 4093898 Local Institution Fukui, Japan, 9188503 Local Institution Kumamoto, Japan, 8628655 Local Institution Miyazaki, Japan, 8800003 Local Institution Nishinomiya-shi, Japan, 6638501 Local Institution Saga, Japan, 8408571 Collapse << |
| NCT01628692 | Hepatitis C Virus | Phase 2 | Completed | - | United States, California ... more >> San Francisco General Hospital San Francisco, California, United States, 94110 Kaiser Permanente Med Ctr San Francisco, California, United States, 94118 United States, Indiana Indiana University Indianapolis, Indiana, United States, 46202 United States, Maryland Johns Hopkins University Lutherville, Maryland, United States, 21093 United States, Tennessee Nashville Medical Research Institute Nashville, Tennessee, United States, 37205 United States, Texas Texas Clinical Research Institute, Llc Arlington, Texas, United States, 76012 United States, Virginia Metropolitan Research Fairfax, Virginia, United States, 22031 Argentina Local Institution Buenos Aires, Argentina, 1119 Local Institution Buenos Aires, Argentina, C1181ACH France Local Institution Creteil Cedex, France, 94010 Local Institution Limoges, France, 87042 Local Institution Marseille Cedex 08, France, 13285 Local Institution Paris Cedex 13, France, 75651 Local Institution Paris Cedex 14, France, 75679 Local Institution Pessac, France, 33600 Local Institution Vandoeuvre Les Nancy, France, 54511 Germany Local Institution Berlin, Germany, 10969 Local Institution Frankfurt, Germany, 60590 Local Institution Hamburg, Germany, 20099 Local Institution Koeln, Germany, 50937 Hungary Local Institution Budapest, Hungary, 1097 Local Institution Budapest, Hungary, 1126 Local Institution Gyula, Hungary, 5700 Spain Local Institution Barcelona, Spain, 08035 Local Institution Madrid, Spain, 28046 Local Institution Valencia, Spain, 46010 Collapse << |
| NCT02032875 | Hepatitis C | Phase 3 | Completed | - | United States, Florida ... more >> University Of Miami Schiff Center For Liver Diseases Miami, Florida, United States, 33136 United States, Michigan University Of Michigan Health System Ann Arbor, Michigan, United States, 48109 United States, Texas Baylor St. Luke'S Medical Center Houston, Texas, United States, 77030 American Research Corporation San Antonio, Texas, United States, 78215 United States, Washington Harborview Medical Center Seattle, Washington, United States, 98104 Collapse << |
| NCT02780362 | Healthy | Phase 1 | Completed | - | Egypt ... more >> Genuine Research Center GRC Cairo, Egypt, 11757 Collapse << |
| NCT02032875 | - | - | Completed | - | - |
| NCT03200184 | Hepatitis C | Phase 4 | Active, not recruiting | May 1, 2018 | Iran, Islamic Republic of ... more >> Shariati Hospital Tehran, Iran, Islamic Republic of, 14117 Collapse << |
| NCT02640157 | Chronic Hepatitis C ... more >> Hepatitis C Virus Genotype 3 Hepatitis C Virus Collapse << | Phase 3 | Completed | - | - |
| NCT02397395 | Renal Impairment ... more >> End-stage Renal Disease Collapse << | Phase 2 | Withdrawn(Trial has been cance... more >>lled due to availability of new therapeutic options for patient population) Collapse << | May 2016 | France ... more >> Paris, France Toulouse Cedex 9, France Villejuif, France Spain Barcelona, Spain Madrid, Spain Santander, Spain Collapse << |
| NCT02496078 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT02640157 | - | - | Completed | - | - |
| NCT03686722 | Diabetes Mellitus, Type 2 ... more >> Hepatitis C Drug Interactions Collapse << | Phase 1 | Completed | - | Egypt ... more >> Drug research centre Cairo, Egypt Collapse << |
| NCT02580474 | Hepatitis C | Phase 4 | Completed | - | Korea, Republic of ... more >> Myeong Jun Song Daejeon, Korea, Republic of Collapse << |
| NCT03490097 | Chronic Hepatitis c ... more >> Metabolic Syndrome Collapse << | Phase 2 Phase 3 | Completed | - | Egypt ... more >> Kobri El koba Armed Forces Hospital Cairo, Egypt Collapse << |
| NCT02556086 | Hepatitis C | Phase 2 | Withdrawn | July 2017 | Canada, Alberta ... more >> Local Institution Edmonton, Alberta, Canada, T6G 2B7 Canada, British Columbia Local Institution Vancouver, British Columbia, Canada, V6Z 2C7 Local Institution Victoria, British Columbia, Canada, V8V 3P9 Canada, Ontario Local Institution Ottawa, Ontario, Canada, K1H 8L6 Local Institution Toronto, Ontario, Canada, M5G 2N2 Canada, Quebec Local Institution Montreal, Quebec, Canada, H4A 3J1 Canada, Saskatchewan Local Institution Regina, Saskatchewan, Canada, S4P 0W5 Canada Local Institution Quebec, Canada, G1V 4G2 France Local Institution Marseille, France, 13009 Local Institution Nantes, France, 44093 Local Institution Nice Cedex, France, 06202 Local Institution Paris Cedex 14, France, 75679 Local Institution Paris Cedex 18, France, 75877 Local Institution Paris, France, 75010 Local Institution Paris, France, 75015 Local Institution Paris, France, 75020 Local Institution Pessac, France, 33604 Collapse << |
| NCT03247296 | - | - | Active, not recruiting | November 10, 2017 | Egypt ... more >> National Hepatology and Tropical Medicine Research Institute Cairo, Egypt, 11796 Collapse << |
| NCT02262728 | Hepatitis C, Chronic | Phase 2 | Completed | - | United States, Texas ... more >> San Antonio, Texas, United States Collapse << |
| NCT01842451 | Chronic Hepatitis C ... more >> CHC HCV Hepatitis C Collapse << | Phase 2 | Completed | - | New Zealand ... more >> New Zealand Auckland, New Zealand New Zealand Christchurch, New Zealand Collapse << |
| NCT03646396 | HCV Coinfection | Not Applicable | Recruiting | December 2025 | Egypt ... more >> Sherief Abd-Elsalam Recruiting Tanta, Egypt Contact: Sherief Abd-elsalam, lecturer 00201000040794 Sherif_tropical@yahoo.com Collapse << |
| NCT02124044 | HIV-HCV | Phase 2 | Completed | - | United States, Maryland ... more >> National Institutes of Health Clinical Center, 9000 Rockville Pike Bethesda, Maryland, United States, 20892 Collapse << |
| NCT02124044 | - | - | Completed | - | - |
| NCT01492504 | - | - | Completed | - | - |
| NCT02565862 | Hepatitis C D... more >>iabetes Mellitus Insulin Resistance Collapse << | Phase 1 | Completed | - | Netherlands ... more >> CRCN, Radboud University Medical Center Nijmegen, Netherlands Collapse << |
| NCT03485846 | Chronic Hepatitis C Genotype 1... more >>b Collapse << | Phase 2 | Active, not recruiting | December 2018 | Russian Federation ... more >> FBIS CSRI of Epidemiology of Federal Service on Customers Moscow, Russian Federation SBEI HPE Moscow State Medical and Dental University n.a. A.I. Evdokimov of Ministry of Health of Russia Moscow, Russian Federation SBHI of Moscow "City Clinical Hospital #24" Moscow, Russian Federation St. Petersburg SBHI Center of Prevention and Fight against AIDS and Infection Diseases Saint Petersburg, Russian Federation Collapse << |
| NCT02596880 | Hepatitis C C... more >>irrhosis Collapse << | Phase 3 | Completed | - | Iran, Islamic Republic of ... more >> Shariati Hospital Tehran, Iran, Islamic Republic of, 14117 Collapse << |
| NCT02309450 | Hepatitis C Virus Genotype 4 I... more >>nfection Collapse << | Phase 2 | Withdrawn(BMS decision) | August 2016 | France ... more >> France All the Regions of the Country, France Collapse << |
| NCT02551861 | Hepatitis C | Phase 2 | Withdrawn | June 2017 | Canada, Alberta ... more >> Local Institution Calgary, Alberta, Canada, T2N 4Z6 Local Institution Edmonton, Alberta, Canada, T6G 2S2 Canada, British Columbia Local Institution Vancouver, British Columbia, Canada, V5Z 1H2 Local Institution Vancouver, British Columbia, Canada, V6Z 2K5 Local Institution Victoria, British Columbia, Canada, V8V 3P9 Canada, Ontario Local Institution Toronto, Ontario, Canada, M5G 2C4 France Local Institution Creteil Cedex, France, 94010 Local Institution Limoges, France, 87042 Local Institution Montpellier Cedex 5, France, 34295 Local Institution Paris Cedex 14, France, 75679 Local Institution Pessac, France, 33604 Local Institution Vandoeuvre Les Nancy, France, 54511 Collapse << |
| NCT02675127 | Healthy | Phase 1 | Completed | - | Egypt ... more >> Genuine Research Center GRC Cairo, Egypt, 11757 Collapse << |
| NCT02032888 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT03487848 | Hepatitis C C... more >>hronic Hepatitis Collapse << | Phase 2 | Recruiting | February 28, 2023 | Australia, Victoria ... more >> Local Institution Recruiting Melbourne, Victoria, Australia, 3052 Contact: Site 0008 Germany Local Institution Recruiting Wuppertal, Germany, 42283 Contact: Site 0005 Poland Local Institution Recruiting Krakow, Poland, 31-202 Contact: Site 0007 Local Institution Recruiting Lodz, Poland, 91-347 Contact: Site 0006 Spain Local Institution Recruiting Barcelona, Spain, 8950 Contact: Site 0003 Local Institution Recruiting Madrid, Spain, 28046 Contact: Site 0009 Taiwan Local Institution Recruiting Taipei, Taiwan, 100 Contact: Site 0002 Collapse << |
| NCT02262728 | - | - | Completed | - | - |
| NCT02032888 | - | - | Completed | - | - |
| NCT02032901 | Hepatitis C | Phase 3 | Completed | - | - |
| NCT02756936 | Healthy | Phase 1 | Completed | - | Egypt ... more >> Genuine Research Center GRC Cairo, Egypt, 11757 Collapse << |
| NCT02624063 | Hepatitis C, Chronic | Phase 4 | Completed | - | Brazil ... more >> Outpatient Clinic of Viral Hepatitis (NUPAIG) São Paulo, Brazil, 04025-001 Collapse << |
| NCT02032901 | - | - | Completed | - | - |
| NCT02268864 | Hepatitis C, Chronic | Phase 2 | Completed | - | Belgium ... more >> Antwerpen, Belgium Brussel, Belgium Gent, Belgium France Creteil, France Lyon, France Montpellier, France Nice N/A, France Paris Cedex 12, France Paris, France Vandoeuvre Les Nancy, France Germany Frankfurt, Germany Hamburg, Germany Hannover, Germany Kiel, Germany Tübingen, Germany Würzburg, Germany Hungary Budapest, Hungary Debrecen, Hungary Spain Badalona, Spain Barcelona, Spain Santander, Spain Valencia, Spain United Kingdom Birmingham, United Kingdom London, United Kingdom Collapse << |
| NCT03369327 | Hepatitis C Virus Infection, R... more >>esponse to Therapy of Human Immunodeficiency Virus Collapse << | Phase 3 | Completed | - | Iran, Islamic Republic of ... more >> Shariati Hospital Tehran, Iran, Islamic Republic of, 14117 Emam Hospital Tehran, Iran, Islamic Republic of Collapse << |
| NCT02268864 | - | - | Completed | - | - |
| NCT02159352 | Hepatitis C | Phase 1 | Completed | - | United States, Texas ... more >> Healthcare Discoveries, Llc D/B/A Icon Development Solutions San Antonio, Texas, United States, 78209 Collapse << |
| NCT01830205 | Hepatitis C | Phase 1 | Completed | - | United States, Florida ... more >> Orlando Clinical Research Center Orlando, Florida, United States, 32809 United States, Minnesota Davita Clinical Research Minneapolis, Minnesota, United States, 55404 Collapse << |
| NCT03063879 | Hepatitis C, Chronic ... more >> Chronic Renal Failure Collapse << | Phase 4 | Active, not recruiting | February 1, 2019 | Iran, Islamic Republic of ... more >> Shariati Hospital Tehran, Iran, Islamic Republic of, 14117 Collapse << |
| NCT03318887 | - | - | Completed | - | France ... more >> Croix-Rousse Hospital, Hospices Civils de Lyon Lyon, France Collapse << |
| NCT03540212 | Chronic HCV Infection | Phase 2 Phase 3 | Recruiting | July 1, 2019 | Egypt ... more >> Pediatric Department, Faculty of Medicine, Ain Shams University Recruiting Cairo, Non-US, Egypt, 11556 Contact: Manal H El-Sayed, MD 00201227461120 mamalhelsayed@yahoo.co.uk Contact: Fatma Soliman E Ebeid, MD 1095569596 ext Ebeid dr.fatma_ebeid@yahoo.com Principal Investigator: Manal H El-Sayed, MD Sub-Investigator: Fatma SE Ebeid, MD Sub-Investigator: Aya M Kamal, MD Sub-Investigator: Mohamed Hassany, MD Sub-Investigator: Mogeb M Saif, MD Sub-Investigator: Samar F Farid Sub-Investigator: Maggie M Abbassi Sub-Investigator: Sara Makkeyah, MD Sub-Investigator: Mary Akhnokh, MD Collapse << |
| NCT01830205 | - | - | Completed | - | - |
| NCT03004625 | Hepatitis C | Phase 3 | Unknown | December 2017 | Taiwan ... more >> Kaohsiung Medical Universsity Recruiting Kaohsiung, Taiwan, 807 Contact: Ming-Lung Yu, MD., PhD. 88673121101 ext 7475 fish6069@gmail.com Contact: Chung-Feng Huang, MD., PhD. 88673121101 ext 7475 fengcheerup@gmail.com Collapse << |
| NCT02865369 | - | - | Not yet recruiting | December 2022 | Korea, Republic of ... more >> Soonchunhyang University Cheonan Hospital Recruiting Cheonan, Chungcheongnam-do, Korea, Republic of, 31151 Contact: SaeHwan Lee, Professor Principal Investigator: SaeHwan Lee, Professor Soon Chun Hyang University Bucheon Hospital Recruiting Bucheon, Gyeonggi do, Korea, Republic of, 14584 Contact: Sang Gyune Kim, Professor 82-32-621-5079 mcnulty@schmc.ac.kr Principal Investigator: Sang Gyune Kim Korea University Ansan Hospital Recruiting Ansan, Gyeonggi-do, Korea, Republic of, 15355 Contact: Young Kul Jung, Professor Principal Investigator: Young Kul Jung, Professor Inha University Hospital Recruiting Jung-gu, Incheon, Korea, Republic of, 22332 Contact: Jin-Woo Lee, Professor Principal Investigator: Jin-Woo Lee, Professor Gachon University Gil Medical Center Recruiting Incheon, Korea, Republic of, 21565 Contact: Oh Sang Kwon, Professor Principal Investigator: Oh Sang Kwon, Professor Severance hospital Recruiting Seoul, Korea, Republic of, 03722 Contact: Jun Yong Park, Professor Principal Investigator: Jun Yong Park, Professor Soonchunhyang University Hospital Recruiting Seoul, Korea, Republic of, 04401 Contact: Jae Young Jang, Professor Principal Investigator: Jae Young Jang, Professor Hanyang university hospital Recruiting Seoul, Korea, Republic of, 04763 Contact: Dae Won Jun, Professor Principal Investigator: Dae Won Jun, Professor Ewha Womans University Mokdong Hospital Recruiting Seoul, Korea, Republic of, 07985 Contact: Tae Hun Kim, Professor Principal Investigator: Tae Hun Kim, Professor Wonju severance christian hospital Recruiting Wonju, Korea, Republic of, 26426 Contact: Moon young Kim, Professor Principal Investigator: Moon young Kim, Professor Collapse << |
| NCT02159352 | - | - | Completed | - | - |

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
